Water For Injection (WFI) Research Grade 100mL Bottle
£10.41
Availability: In stock
Water for Injection (WFI) – Cell Culture Grade
Description:
WFI for Cell Culture is a high‑purity, cell‑culture‑grade water produced through a 4‑stage distillation process and sterilized using purified water systems. It meets the stringent quality requirements for sterile Water for Injection as defined in USP and EP monographs. Manufactured in an ISO‑certified, cGMP‑compliant facility, this product provides exceptional purity and safety for biopharmaceutical and laboratory workflows.
This WFI meets the highest standards required in biopharmaceutical, pharmaceutical, and diagnostic industries, and is widely used across biochemistry, molecular biology, cell biology, chemistry, physiology, and pathology. It is especially suited for the preparation of high‑purity reagents, buffers, and media in research laboratories.
Key Features
- Grade: Cell culture–grade, WFI‑quality
- Purification method: 4‑stage distillation
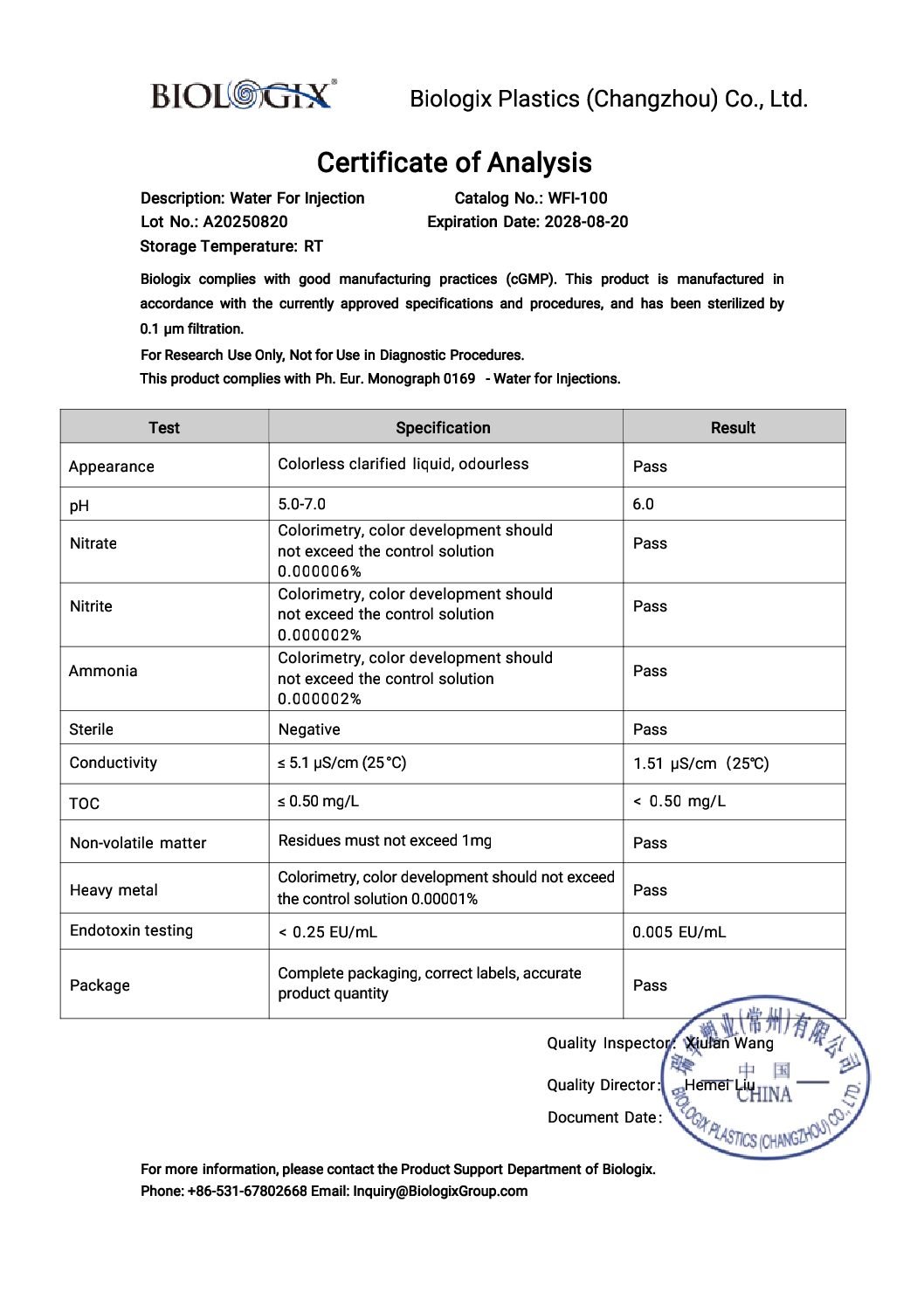
- Sterility: Sterile; meets USP/EP requirements for Water for Injection
- Manufacturing: Produced in an ISO‑certified, cGMP facility
- Endotoxin level: Ultra‑low (suitable for sensitive biological work)
- Particulate level: Meets WFI particulate specifications
- Use case: Ideal for preparation of high‑purity laboratory solutions
Applications
Used across a wide range of scientific fields, including:
- Biochemistry
- Molecular biology
- Cell biology and tissue culture workflows
- Physiology and pathology research
- Analytical chemistry and buffer preparation
- General laboratory reagent preparation
Suitable for:
- Preparation of sterile reagents and buffers
- Media formulation
- Cleaning and rinsing of sensitive equipment (research‑only)
- High‑purity solution preparation
Storage & Stability
- Storage temperature: Ambient or as required by SOP
- Packaging: Sterile, sealed bottles
- Handling: Use aseptic technique after opening
- Shelf life: Defined by batch COA
Quality & Compliance
- Manufactured under cGMP
- Produced in an ISO‑certified facility
- Meets USP and EP monograph specifications for WFI
- Every lot supplied with Certificate of Analysis (COA)
Intended Use
For laboratory research use only.
Not for human or veterinary use.
Not for clinical, diagnostic, or therapeutic applications.
Use appropriate laboratory safety procedures.
Search‑Optimised Keywords
WFI, Water for Injection, cell‑culture‑grade water, sterile WFI, USP WFI, EP WFI, biopharmaceutical‑grade water, molecular biology water, high‑purity laboratory water, cGMP water, ISO‑certified WFI, distilled sterile water for research.
| Weight | 100 g |
|---|